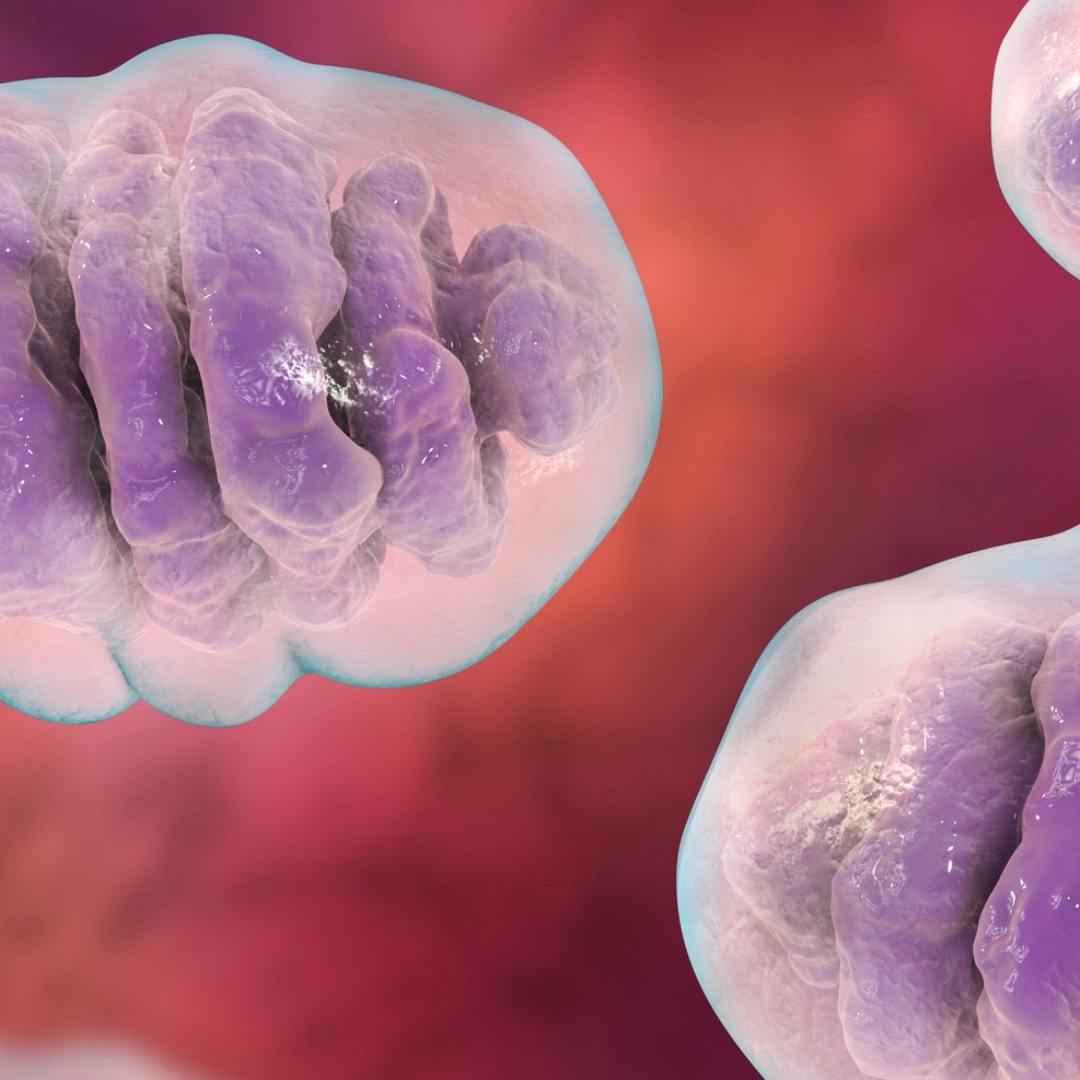
Transcranial photobiomodulation (tPBM) — the application of near-infrared light to the skull and underlying brain tissue — is an emerging field with a growing body of preclinical and clinical evidence supporting effects on neurological function, cognitive performance, and neuroprotection. Unlike visible red light, near-infrared wavelengths (800–1100 nm) penetrate the scalp, skull, and meninges to reach the cerebral cortex. Cytochrome c oxidase in neurons is the primary chromophore, and photon absorption increases mitochondrial ATP production, reduces neuroinflammation, and stimulates neuroprotective signaling pathways including BDNF and NGF upregulation.
Preclinical evidence from traumatic brain injury (TBI), stroke, and neurodegeneration models is extensive and mechanistically compelling. Human clinical trials, though earlier in development than musculoskeletal applications, demonstrate measurable effects on cognitive function, mood, and cerebral blood flow. Pilot RCTs in TBI survivors show improvements in cognitive speed, memory, and PTSD symptom scores. Studies in healthy adults show enhanced executive function, working memory, and reaction time following single sessions of tPBM at 1064 nm or 810 nm. The prefrontal cortex appears particularly responsive, likely due to its high metabolic demand and proximity to the skull surface.
Emerging clinical applications include major depressive disorder, PTSD, mild cognitive impairment (MCI), Alzheimer's disease, and Parkinson's disease. While most human trials remain in Phase I/II stages with small sample sizes, the mechanistic rationale is strong — mitochondrial dysfunction is increasingly recognized as a core pathophysiology in neurodegenerative diseases, and PBM's ability to restore mitochondrial function non-invasively positions it as a potentially transformative neuroprotective intervention. Larger multi-center RCTs are now underway.